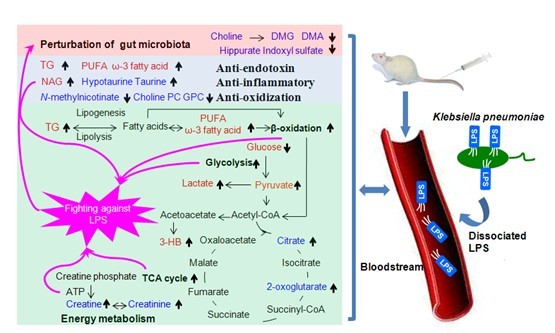
Bacteremia, the presence of viable bacteria in the blood stream, is often associated with several clinical conditions. Bacteremia can lead to multiple organ failure if managed incorrectly, which makes providing suitable nutritional support vital for reducing bacteremia-associated mortality. Bacteremia is expected to generate measurable changes in metabolic levels. Therefore it is possible to monitor dynamic metabolic changes associated with bacteremia and identify metabolites related to the event. Developing an in-depth and systematic study of changes associated with bacteremia could provide a comprehensive view on the host metabolic response to bacteremia and open a window for nutritional intervention against the disease.
In order to provide such information, Prof. Yulan Wang’s group investigated the metabolic consequences of a Klebsiella pneumoniae (K. pneumoniae) infection in vivo by employing a combination of 1H nuclear magnetic resonance spectroscopy and multivariate data analysis. K. pneumoniae was intravenously infused in rats; urine and plasma samples were collected at different time intervals. It was found that K. pneumoniae-induced bacteremia stimulated glycolysis and the tricarboxylic acid cycle and also promoted oxidation of fatty acids and creatine phosphate to facilitate the energy-demanding host response. In addition, K. pneumoniae bacteremia also induced anti-endotoxin, anti-inflammatory and anti-oxidization responses in the host. Furthermore, bacteremia could cause a disturbance in the gut microbiotal functions as suggested by alterations in a range of amines and bacteria-host co-metabolites. Their results suggest that supplementation with glucose and a high-fat and choline-rich diet could ameliorate the burdens associated with bacteremia. Their research provides underlying pathological processes of bacteremia and a better understanding of the clinical and biochemical manifestations of bacteremia. The result has been published online in PLoS ONE (http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0051060).
This work is funded by the Ministry of Science and Technology of China, the National Nature Science Foundation of China and Chinese Academy of Sciences.




